Australia’s Therapeutic Goods Administration (TGA) has approved the Novavax coronavirus vaccine for use in adults.

But you might be wondering, why do we need another vaccine at all when most of us are fully vaccinated and some have received a booster?
The government says in part it’s about offering more choice to those who are yet to be vaccinated — and it’s not yet clear whether the shots will be used as boosters.
What is the Novavax vaccine? Is it different?
The Novavax vaccine works in a different way to other vaccines that protect against infection or severe symptoms of COVID-19.
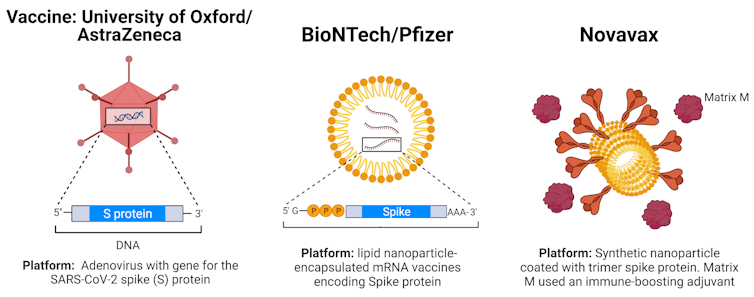
The more common mRNA vaccines like Pfizer and Moderna contain a genetic instruction telling your body how to make a spike protein like that on the SARS-CoV-2 virus, so that your immune system learns to recognise and fight it.
Novavax, on the other hand, delivers the spike protein itself into your body, and your immune system then learns to fight the virus in the same way.
Novavax has not been as widely taken up around the world as other vaccines, but the Australian government has had an order for 51 million doses in place for more than a year.
Two separate worldwide trials found Novavax had a 90 per cent efficacy rate at preventing symptomatic infection with no major signals of adverse events.
But those trials were conducted before the emergence of the Omicron variant. The company says it is conducting further studies on the efficacy of the vaccine against newer variants.
Why are we approving a new vaccine now? When will Novavax be available?
MRNA vaccines and viral vector vaccines such as the AstraZeneca shot have both been shown to be safe and effective at protecting people against COVID-19.
However, TGA head John Skerritt acknowledged a very small section of the community had been hesitant about those vaccines in particular, and expressed a preference for the Novavax formula.
“There are some individuals who have been waiting for Novavax and it’s great that it’s finally been approved.” he said.
“Our dream is that we might turn our 95 per cent [vaccination rate] into 97 or 98 per cent in this country. Who knows.”
Paul Griffin, the director of infectious diseases at Mater Health, was the principal investigator in early Novavax trials.
He said while other vaccines were completely safe and effective, providing more choice to people who had not taken them was the right thing to do.
“For a variety of reasons, it seems some have remained reluctant to receive these vaccines thus far so an additional option, based on what is considered perhaps a more traditional platform, is likely to increase our vaccination rate even further,” he said.
Novavax still has to get final approval from the Australian Technical Advisory Group on Immunisation (ATAGI), which will decide who the vaccine will be made available to.
The government says it has plans to distribute the vaccine by the end of February.
“It will be made available in the coming weeks to supply coming into the country to be distributed through states and territories and general practices and pharmacies that seem to order it,” Health Minister Greg Hunt said.
Will Novavax be used for boosters? What about kids?
Currently the TGA approval is only for a two-dose initial vaccination course in people aged over 18.
But that could change.
“I know there is interest in the potential of Novavax being used as a booster, or even in adolescents or as a paediatric dose,” Professor Skerritt said.
“The company hasn’t yet submitted an application to us for that, but we’re talking to them.”
What is the dose interval for Novavax? Are there side effects?
The TGA has approved Novavax for use in a two-dose course, with the two jabs being administered 21 days apart.
One phase three trial on Novavax found its most commonly reported side effects were headaches, muscle pain and fatigue.
In the study, side effects were more commonly reported after the second dose of the vaccine.


Has anyone for the most brief moment stopped to ask yourself: After their alleged SARS-CoV2, aka ‘covid-19’ FDA approval of Comitnaty vaccination in August 2021, why on God’s green earth would there be any need for producing some another alleged ‘covid-19 vaccination’?
Hmmm…maybe there’s some underlying ‘but highly secret reason’ people are not being told about?
If I could…I’d 100% straight-forward ask Moderna, BioNTech, Phizer,J&J ad nauseam…after a six month wait…does fear or good sense hold you back from producing the first dose of Comirnaty?
Or should I ask why there’s a recently released tablet form…or Novavax.
When there’s one plus one adding up to 10 1/5th…there’s something very fishy going on.
If someone wants fish, why would they be given a serpent? Matthew chapter 7 verse 10.
(0_o)
If they say other vaccines are perfectly safe, we cannot trust that this new one is safe too.
I hope you are wrong Alex