STORY AT-A-GLANCE
- In November 2021, Brook Jackson, a whistleblower who worked on Pfizer’s Phase 3 COVID jab trial in the fall of 2020, warned she’d seen evidence of fraud in the trial
- With the release of Pfizer trial data — which they tried to withhold for 75 years — additional problems suggestive of fraud and data manipulation are coming to light
- Trial site 1231, located in Argentina, somehow managed to recruit 10% of the total trial participants, 4,501 in all, and they did so in just three weeks, and without a contract research organization — a feat that has many questioning whether fraud was committed
- The lead investigator for trial site 1231 is Dr. Fernando Polack, who also happens to be a consultant for the U.S. Food and Drug Administration’s Vaccines and Related Biological Products Advisory Committee (RBPAC), a current adjunct professor at Vanderbilt University in Tennessee, an investigator for Fundación Infant, funded by the Bill & Melinda Gates foundation, and the first author of Pfizer’s paper, “Safety and Efficacy of the BNT162b2 mRNA COVID-19 Vaccine,” published at the end of December 2021
- Site 1231 held a second enrollment session, given the designation of “site 4444.” The 4444 trial site data raise another red flag. It supposedly enrolled 1,275 patients in a single week, from September 22 through 27, 2020 — the last week that recruitment could take place to meet the data cutoff for the FDA meeting in December 2020. Was “site 4444” fabricating data to create the appearance that the jab was having an effect?
In November 2021, Brook Jackson, a whistleblower who worked on Pfizer’s Phase 3 COVID jab trial in the fall of 2020, warned she’d seen evidence of fraud in the trial.
Data were falsified, patients were unblinded, the company hired poorly trained people to administer the injections, and follow-up on reported side effects lagged way behind. The revelation was published in The British Medical Journal. In his November 2, 2021, report, investigative journalist Paul Thacker wrote:1
“Revelations of poor practices at a contract research company helping to carry out Pfizer’s pivotal COVID-19 vaccine trial raise questions about data integrity and regulatory oversight …
[F]or researchers who were testing Pfizer’s vaccine at several sites in Texas during that autumn, speed may have come at the cost of data integrity and patient safety … Staff who conducted quality control checks were overwhelmed by the volume of problems they were finding.”
Jackson, a former regional director of Ventavia Research Group, a research organization charged with testing Pfizer’s COVID jab at several sites in Texas, repeatedly “informed her superiors of poor laboratory management, patient safety concerns and data integrity issues,” Thacker wrote.
When her concerns were ignored, she finally called the U.S. Food and Drug Administration and filed a complaint via email. Jackson was fired later that day after just two weeks on the job. According to her separation letter, management decided she was “not a good fit” for the company after all.
She provided The BMJ with “dozens of internal company documents, photos, audio recordings and emails” proving her concerns were valid, and according to Jackson, this was the first time she’d ever been fired in her 20-year career as a clinical research coordinator.
BMJ Report Censored
Disturbingly, social media actually censored this BMJ article and published pure falsehoods in an effort to “debunk” it. Mind you, the BMJ is one of the oldest and most respected peer-reviewed medical journals in the world! The Facebook “fact check” was done by Lead Stories, a Facebook contractor, which claimed the BMJ “did NOT reveal disqualifying and ignored reports of flaws in Pfizer’s” trials.2
In response, The BMJ slammed the fact check, calling it “inaccurate, incompetent and irresponsible.”3,4,5 In an open letter6 addressed to Facebook’s Mark Zuckerberg, The BMJ urged Zuckerberg to “act swiftly” to correct the erroneous fact check, review the processes that allowed it to occur in the first place, and “generally to reconsider your investment in and approach to fact checking overall.” As noted by The BMJ in its letter, the Lead Stories’ fact check:7
- Inaccurately referred to The BMJ as a “news blog”
- Failed to specify any assertions of fact that The BMJ article got wrong
- Published the fact check on the Lead Stories’ website under a URL that contains the phrase “hoax-alert”
Pfizer Trial Data Raises Suspicions of Fraud
Now, with the release of Pfizer trial data8 — which they tried to withhold for 75 years — internet sleuths are finding additional problems suggestive of fraud and data manipulation. May 9, 2022, a Twitter user named Jikkyleaks posted a series of tweets questioning data from Pfizer trial sites 1231 and 4444.9
Trial site 1231, located in Argentina, somehow managed to recruit 10% of the total trial participants, 4,501 in all, and they did so in just three weeks, and without a contract research organization (CRO). CROs like the Ventavia Research Group, which Jackson worked for, provide clinical trial management services. The lead investigator for trial site 1231 is Dr. Fernando Polack,10 who also happens to be:11
- A consultant for the U.S. Food and Drug Administration’s Vaccines and Related Biological Products Advisory Committee (RBPAC) since 2017
- A current adjunct professor at Vanderbilt University in Tennessee
- An investigator for Fundación Infant,12 which is funded by the Bill & Melinda Gates foundation13
- The first author of Pfizer’s paper,14 “Safety and Efficacy of the BNT162b2 mRNA COVID-19 Vaccine,” published at the end of December 2021
As noted by Jikkyleaks, Polack “is literally the busiest doctor on the planet,” because in addition to all those roles, he also managed to single-handedly enroll 4,500 patients in three weeks, which entails filling out some 250 pages of case report forms (CRFs) for each patient. That’s about 1,125,000 pages total. (CRFs are documents used in clinical research to record standardized data from each patient, including adverse events.)
This recruitment also took place seven days a week, which is another red flag. “Weekend recruitment for a clinical trial would be odd. Staff are needed to fill out that many record forms (CRFs) and there are potential risks to the trial, so you need medical staff. It would be highly unusual,” Jikkyleaks notes.
Is Polack just a super-humanly efficient trial investigator, or could this be evidence of fraud? As noted by Steve Kirsch in the featured video and an accompanying Substack article,15 Polack is the coordinator for a network of 26 hospitals in Argentina, so perhaps it’s possible he could have recruited 57 patients per week per hospital, but it seems highly unlikely.
Questions Surround Site 4444 Data
Now, “site 4444” does not exist. It’s actually the same as site 1231. It appears site 1231 held a second enrollment session, and these were for some reason given the designation of 4444. The 4444 trial site data raise another red flag.
Site 4444 (the second enrollment session for site 1231) supposedly enrolled 1,275 patients in a single week, from September 22 through 27, 2020, and the suspicious thing about that — aside from the speed — is the fact that this was the last week that recruitment could take place to meet the data cutoff for the FDA meeting in December 2020. Jikkyleads writes:16
“My guess: they needed enough numbers of ‘positive PCR tests’ in the placebo group to show a difference between groups for that VRBPAC meeting on the 10th Dec, and they didn’t have them. So, site 4444 appeared and gave them their ‘perfect’ result. Bravo.”
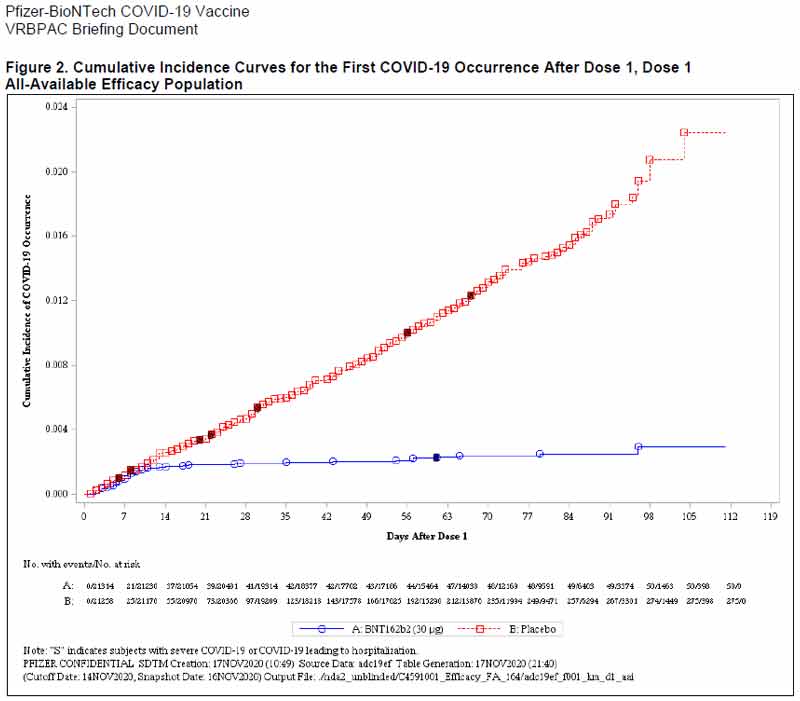
Kirsch notes:17
“Was there fraud in the Pfizer trial? Without a doubt. The story of Maddie de Garay is a clear case of that. Brook Jackson has evidence of fraud; she has 17 lawyers working for her. If there wasn’t fraud, these lawyers wouldn’t be wasting their time.
This new data on Site 1231/4444 looks suspicious to me. It looks too good to be true. But we can’t make the call without more information. Undoubtedly, the mainstream media will not look into this, Pfizer will remain silent, and Polack will be unreachable for comment. The lack of transparency should be troubling to everyone. That is the one thing we can say for sure.”
Pfizer Documents Reveal COVID Jab Dangers
Among the tens of thousands of Pfizer documents released by the FDA so far, we now also have clear evidence of harm. For nurse educator John Campbell, featured in the video above, these documents appear to have served as a “red pill,”18 waking him up to the possibility that the jabs may indeed be far more dangerous than anyone expected, including himself.
In the video, Campbell reviews the documents listed as “5.3.6. Postmarketing Experience,” which were originally marked “confidential.” They reveal that, cumulatively, through February 28, 2021, Pfizer received 42,086 adverse event reports, including 1,223 deaths.
To have 1,223 fatalities and 42,086 reports of injury in the first three months is a significant safety signal, especially when you consider that the 1976 swine flu vaccine was pulled after only 25 deaths.
As noted by Campbell, “It would have been good to know about this at the time, wouldn’t it?” referring to the rollout of the jabs. Campbell has been fairly consistent in his support of the “safe and effective” vaccine narrative, but “This has just destroyed trust in authority,” he said.
158,000 Recorded Side Effects — A World Record?
The first really large tranche of more than 10,000 Pfizer documents was released March 1, 2022. (You can find them all on PHMPT.org.19)
In this batch were no less than nine single-space pages of “adverse events of special interest,” listed in alphabetical order20 — 158,000 in all!
The first side effect on this shockingly exhaustive list is a rare condition known as 1p36 deletion syndrome, which results in severe intellectual disability, seizures, vision problems, hearing loss, breathing problems, brain anomalies, congenital heart defects, cardiomyopathy, renal anomalies, genital malformation, metabolic problems and more.
To see the first page, click the link below. The first side effect on this shockingly exhaustive list is a rare condition known as 1p36 deletion syndrome. This condition, caused by the deletion of DNA in chromosome 1p36, results in developmental delays, severe intellectual disability, seizures, vision problems, hearing loss, breathing problems, brain anomalies, congenital heart defects, cardiomyopathy, renal anomalies, genital malformation, metabolic problems and more.21,22
Life expectancy depends on the amount of DNA that has been deleted. This, at bare minimum, sounds like something a pregnant woman might want to know before she gets the shot.
CRF Anomalies Raise Questions of Fraud
After reviewing some of the released CRFs in the March 1 tranche, investigative journalist Sonia Elijah also discovered several problems, including the following:23
| Patients entered into the “healthy population” group who were far from healthy — For example, one such “healthy” participant was a Type 2 diabetic with angina, a cardiac stent and a history of heart attack. |
| Serious adverse event (SAE) numbers were left blank — Ventavia site No. 1085 has a particularly large number of missing SAE numbers. |
| Missing barcodes for samples collected — Without those barcodes, you can’t match the sample to the participant. |
| Suspicious-looking SAE start and end dates — For example, the so-called “healthy” diabetic suffered a “serious” heart attack October 27, 2020. The “end” date is listed as October 28, the next day, which is odd because it was recorded as serious enough to require hospitalization.Also, on that same day, October 28, the patient was diagnosed with pneumonia, so likely remained hospitalized. “This anomaly raises doubt as to the accuracy of these recorded dates, potentially violating ALOCA-C clinical site documentation guidelines for clinical trials,” Elijah writes. |
| Unblinded teams were responsible for reviewing adverse event reports for signs of COVID cases, and to review severe COVID cases — Yet in some cases they appear to have dismissed the possibility of an event being COVID-related, such as pneumonia. This despite the fact that Pfizer’s protocol (section 8.2.4) lists “enhanced COVID-19” (i.e., antibody dependent enhancement) as a potential side effect to be on the lookout for. As noted by Elijah:“Inadvertently, this could have led to bias, as the unblinded teams would have been aware which participants were assigned the placebo and those who received the vaccine. They might have been under pressure by the sponsor for the trial to go a certain way and for events like ‘COVID Pneumonia’ to be classified simply as pneumonia.” |
| Impossible dating — The diabetic who suffered a heart attack followed by pneumonia (which may have been unacknowledged COVID pneumonia) died, and the date of death is listed as the day before the patient supposedly went for a “COVID ill” visit.Clearly, it’s impossible for a dead person to attend a medical visit, so something is wrong here. The clinical investigator note states: “There cannot be a date later than date of death. Please remove data from the COVID illness visit and add cough and shortness of breath as AEs (adverse events).” “What kind of pressure was being exerted here?” Elijah asks. |
| Second dose administered outside the three-week protocol window. |
| Observation period appears to have been an automatic entry — According to the protocol, each participant was to be observed by staff for a minimum of 30 minutes.A majority of the CRFs state 30 minutes, which raises the question: Were participants observed for adequate amounts of time, or did they simply put down “30 minutes” as an automatic entry? Why is there so little variety in the observation times? If participants were not adequately observed, their safety was put at risk, which was one of Jackson’s concerns. |
| Adverse events listed as “not serious” despite extended hospital stay — In one case, the participant fell and suffered facial lacerations the day after the second dose and was hospitalized for 26 days, yet the fall was not reported as serious.Other anomalies in this particular case include listing the fall as being caused by a “fall” unrelated to the study treatment, and the facial laceration being the result of “hypotension” (low blood pressure). The SAE number is also missing for the facial lacerations.Elijah writes, “Doubts can be raised over the credibility of this information given the fall and facial lacerations were intrinsically related. So, if facial lacerations were due to ‘hypotension’ then the fall should be due to that too.” Might low blood pressure be an effect of the experimental shot? Possibly. Especially when you consider the patient fell the day after being given the second dose.Even more suspicious: the causality for the fall was recorded as “related” (to the treatment) on the serious adverse event form, but listed as “not related” on the adverse event CRF. A note states, “Please confirm correct causality.” |
| Dismissing brand new health problems as unrelated to the treatment — For example, in one case, a female participant with no medical history of impaired kidney function was diagnosed with kidney stones and severe hypokalemia, requiring hospitalization, one month after her second dose. Yet despite her having no history of kidney problems, both events were dismissed as “not related” to the study treatment and no further investigation was done. |
In closing, Elijah wrote:24
“All the evidence gleaned over a limited time appears to back up whistleblower Jackson’s claims of poor trial site data management and raises questions as to how Ventavia conducted the Pfizer clinical trials.
The errors and anomalies in the CRFs also allude to her claims that the clinical research associates were not trained adequately, with many having had no prior clinical experience history. If such egregious findings are true at these sites, could they manifest at other trial sites around North America and beyond?”
Can You Trust Pfizer?
Pfizer, which was quickly given emergency use authorization (EUA) for its COVID-19 mRNA gene therapy shot, has a long list of criminal verdicts against it:
| In 2002, Pfizer and two subsidiaries paid $49 million to settle civil claims that it had failed to report best prices for its drug Lipitor, as is required under the Medicaid Drug Rebate Statute.25 |
| In 2004, a Pfizer subsidiary Warner-Lambert pleaded guilty and paid more than $430 million to settle criminal charges and civil liability from fraudulent marketing practices.26 |
| In 2007, another subsidiary was found guilty of paying out kickbacks for formulary placement of its drugs and had to pay a fine of $34 million.27 |
| Two years later, in 2009, Pfizer was found guilty of health care fraud and ordered to pay the largest penalty ever for this kind of offense.28 When announcing the record penalty of $2.3 billion against the drug giant, the U.S. Department of Justice said one of the charges was a felony. The other charges stemmed from false actions and false claims submitted to federal health care programs. |
| In 2010, the company was again ordered to pay $142 million in damages for fraudulent marketing and promoting the drug Neurontin for unapproved uses.29 |
| Less than 10 years later, in 2018, Pfizer was again caught in an illegal kickback scheme and agreed to pay $23.8 million to resolve claims that it used a foundation as a conduit to pay the copays of Medicare patients taking three of its drugs.30 |
As noted in the journal Healthcare Policy in 2010,31 “Pfizer has been a ‘habitual offender,’ persistently engaging in illegal and corrupt marketing practices, bribing physicians and suppressing adverse trial results.” The article also highlights the crimes of Johnson & Johnson, another COVID jab maker.
Despite its tarnished history, we’re now expected to trust that everything Pfizer does is above-board. I don’t think so. A company that continues getting caught committing the same crimes over and over again clearly has a deeply established ethical rot within its corporate structure that fines simply have no effect over.
Has Pfizer committed fraud in its COVID jab trials as well? It sure looks that way. Time will tell whether attorneys will have enough for a conviction in the future. If fraud did take place, Pfizer can (and likely will) be held liable for the more than one million injuries its injection has caused in the U.S. alone, and we all look forward to that reckoning.


